Turning Off The Lights On Malaria
Malaria is responsible for about 800,000 deaths a year. In order to control the disease genetically modified mosquitoes have been created that can no longer harbor the plasmodium that transmits malaria. If these plasmodium resistant mosquitoes were ever released into the wild they will be out numbered by the billions of wild mosquitoes that buzz around India and equatorial Africa. To have any effect on the spread of malaria they will have to out compete the wild type blood suckers. The genetic changes responsible for their resistance have to rapidly spread through the mosquito population.
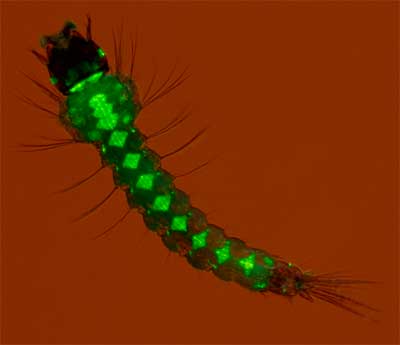
Prof. Andrea Crisanti of Imperial College, who also created the mosquito larva with fluorescent testicles, has used selfish genes in a proof of concept experiment that demonstrates how the gene necessary for housing plasmodia in the mosquito can quickly and thoroughly be disrupted in a scrounge of mosquitoes. In an April 2011 Nature article Crisanti and co-workers describe how they created a selfish gene that targeted and destroyed the GFP gene in the mosquito’s sperm. Since the selfish gene sneaks into the genome of all the mosquito offspring it spreads very quickly. To test their ideas they created a population of GFP expressing mosquitoes, see image. The fluorescent glow of the green critters was then gradually turned off by the release of a small number of mosquitoes with the GFP targeting selfish genes. After 12 generations over 60% of the GFP genes were compromised. When larger populations with the selfish gene were released the GFP fluorescence was extinguished even faster. According to Crisanti, “The next step is to make the selfish gene break up not the fluorescent protein gene but one that is crucial for malaria transmission. It could be an odor-recognition gene that helps the mosquito find its host, for instance, or one that the malaria parasite needs to enter the mosquito’s salivary glands; the team already has 10 to 15 candidates.”
Update: 2019 Crisanti lab uses gene drives to try and eradicate malaria.